ePrescribe: Get Approved for EPCS
In order to prescribe controlled substances, you need at least one supporting user in ePrescribe to approve your identity. This is a rule from the DEA.
You should have been asked to include this user upon ePrescribe sign up, but if you do not have a supporting user yet, invite one with the directions below. Please note that you cannot add yourself (the Prescriber) as the supporting user.
Are you a solo practitioner who needs to add a Supporting User to be approved for EPCS, but you do not want to add them as staff in PracticeQ? If so, please use this article: Invite a Supporting User for EPCS Approval.
First, add the user in PracticeQ:
- If the user is already in PracticeQ, skip to the next section.
- Click More → Team.
- If you wish to add the user as an Assistant, stay in the Assistants tab. If you wish to add the user as a Practitioner, select the Practitioners tab.
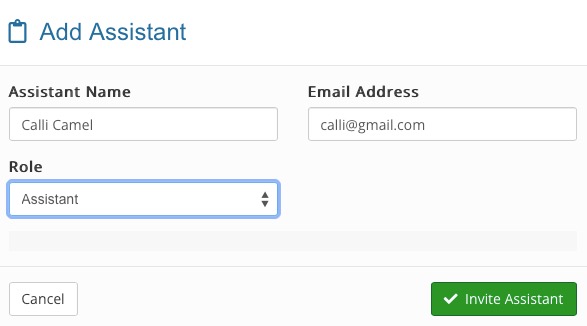
- Click + Add New Assistant.
- Enter the user's name, email, and role.
- Click Invite Assistant.
- Ask the user to open their invitation email and set up their PracticeQ account.
Note: It is free to add an assistant in PracticeQ; for this reason, most practitioners add supporting users as assistants.
Next, invite them as a Supporting User in ScriptSure:
Supporting users can view the Prescriptions tab on clients and queue up medications for practitioner approval.
- Click More → ePrescribe.
- Scroll down to the Practitioners or Assistants heading.
- If you do not see the user you just added, log out of PracticeQ and log back in.
- Choose whether you want the invitee to be a Basic Administrator or a Full Administrator for ePrescribe.
- Basic Administrator:
- User is permitted to change practice and clinical application settings
- Full Administrator:
- Check if this user is permitted to Add, Invite and Edit users of the organization
- If you are inviting a practitioner but you do not want them to prescribe, uncheck the Prescriber checkbox before sending the invite. This checkbox is only used for invitations. You cannot control whether they are a prescriber or not after the invitation is sent.
- Click the blue Send Invitation Email button. This will send them an email to sign up.
‼️IMPORTANT: You need at least one other user set as a Full Administrator to approve you for EPCS the first time. If you select None, the user will be set up as a Basic Administrator. Making the user a Full Administrator in ePrescribe will not grant them admin permissions in PracticeQ!
Steps for Supporting User
If you were already a user in PracticeQ, complete the following steps:
- Click Lists → Clients.
- Open the profile of a client who is enrolled in eRx. You do not have to prescribe a medication for the client, so it does not matter which client you choose.
- Select the clients Prescriptions tab.
- The ScriptSure interface will open.
- You should see a prompt to approve the access request. If you do not see this prompt, reach out to hello@intakeq.com.
- Once you have approved the Provider, you should not need to do it again.

If you were not yet already a user in PracticeQ, complete the following steps:
- Open the invitation email from ScriptSure and click Setup Account.
- Create your password and log in to ScriptSure.
- Click Add User at the top right.
- A pop-up that shows EPCS Request will appear. Click Review and then Approve next to the name of the Provider.
- Once you have approved the Provider, you should not need to do it again.
DEA's Second User (Administrator) Requirement
The Controlled Substances Act (CSA), 21 U.S.C. 801-904, prevents the diversion of controlled substances into improper channels by requiring that controlled substances only be prescribed by practitioners registered with DEA (or exempt from the registration requirement).
Thus, one of DEA's primary goals in the IFR was to ensure that non-registrants cannot improperly gain access to electronic prescription applications— i.e., the computer software practitioners use to electronically issue their prescriptions. Obviously, if non-registrants could gain access to these applications, they might be able to use them to fraudulently generate or alter electronic prescriptions for controlled substances, thereby diverting these controlled substances in violation of the CSA.
Thus, the IFR contains a number of measures designed to minimize, to the greatest extent possible, the potential for the diversion of controlled substances through such misuse of electronic prescription applications. These include the IFR's approaches to identity proofing (verifying that the user of an electronic prescription application is who he or she claims to be) and logical access control (verifying that the authenticated user has the authority to perform the requested action).
Under the IFR, a practitioner can only sign and issue an electronic prescription by using an authentication credential, and a practitioner can only receive such a credential after having his or her identity verified. For individual practitioners (as opposed to practitioners associated with an institutional practitioner registrant), such identity proofing is done by authorized third parties that, after verifying a registrant's identity, issue an authentication credential to the registrant. These third parties must be federally approved credential service providers (CSPs) or certification authorities (CAs).
Ultimately, the second user logs in once to approve the provider for prescribing. Once the approval is complete, the prescriber can prescribe and the second user does not need to log in again.
.png)
